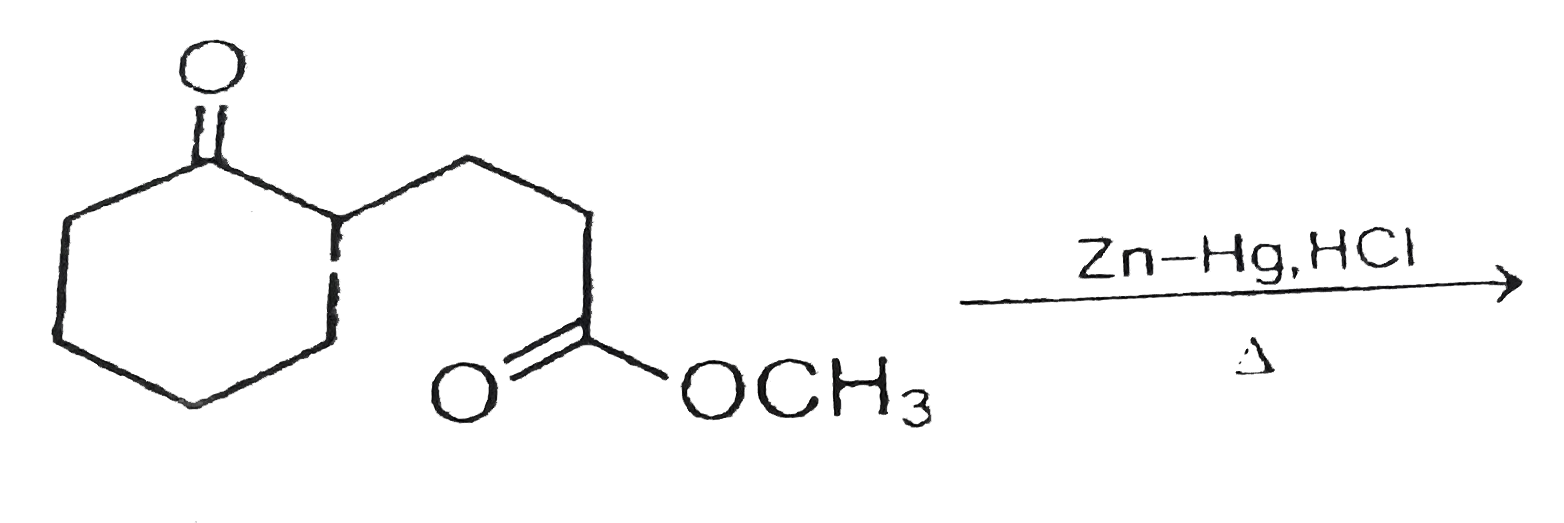
Treatment of benzaldehyde (C_6H_5CHO) with Zn(Hg) in aqueous HCl forms a compound Z that has a molecular ion at 92 in its mass spectrum. Z shows absorptions at 3150-2950, 1605, and 1496
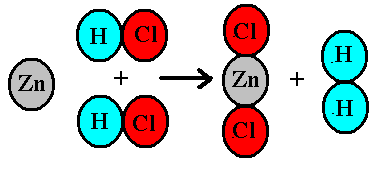
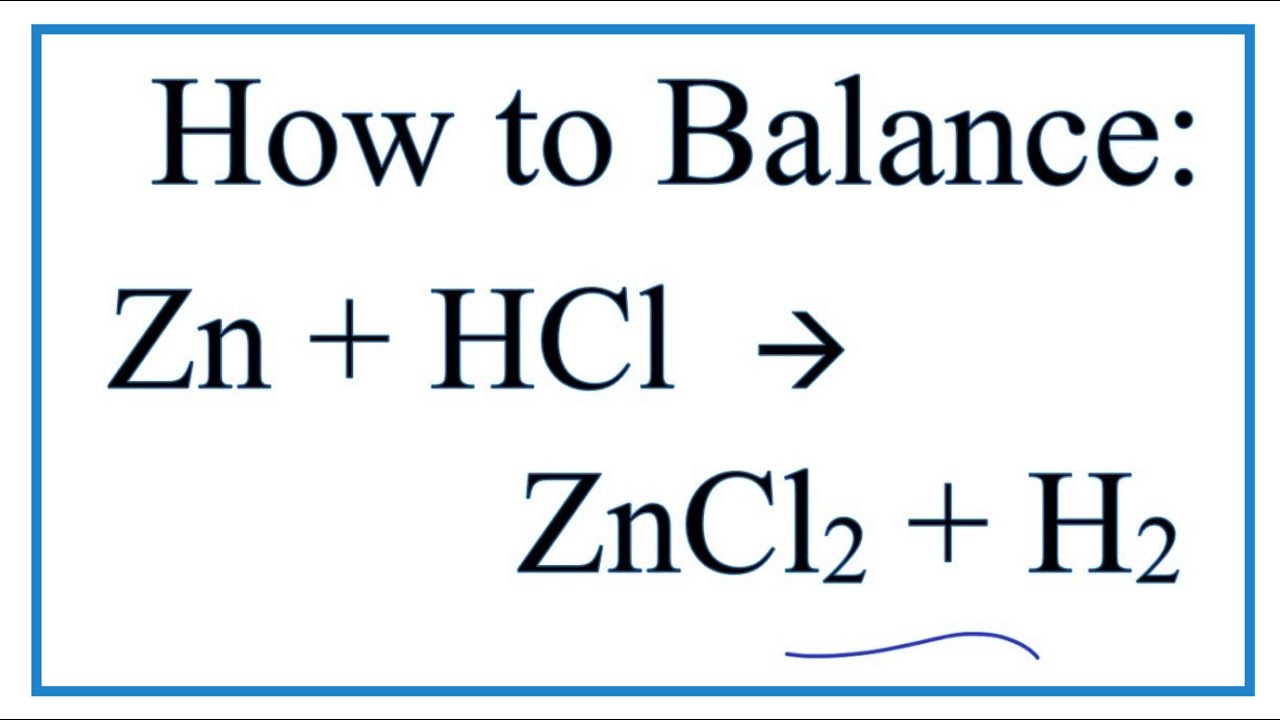
In the reaction Zn + 2HCl -> ZnCl_2 + H_2, how many moles of hydrogen will be formed when 4 moles of HCl are consumed? | Socratic
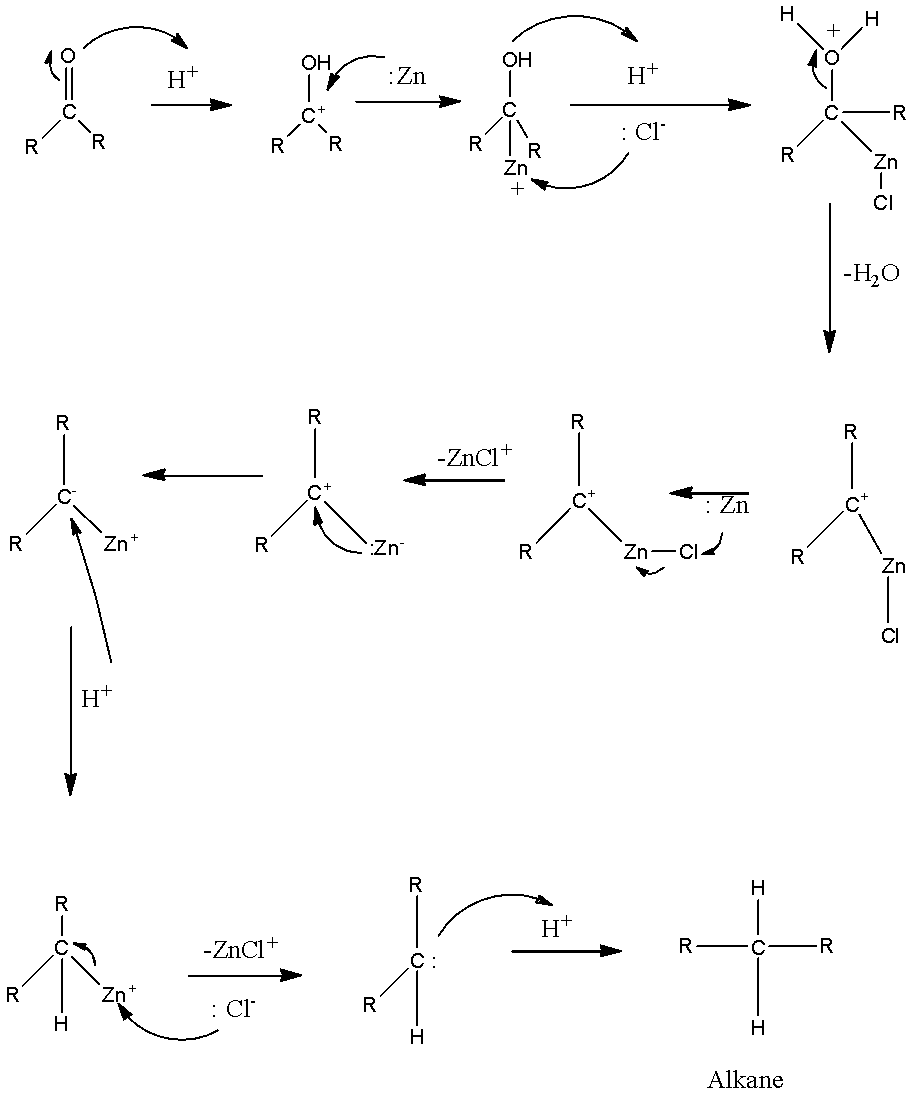
Which of the following is reduced with Zn-Hg and HCl to give alkane?(a).Ethyl acetate(b).Acetic acid(c).Acetamide(d).Butan-2-one

SOLVED: Consider the reaction between zinc metal and hydrochloric acid. Which of the following would increase the rate of the reaction? 3. ZnCl2 2. HCl Zn H2 using zinc powder instead of

How to Balance H2SO4 + Zn = ZnSO4 + H2 (Sulfuric Acid + Zinc) | How to Balance H2SO4 + Zn = ZnSO4 + H2 (Sulfuric Acid + Zinc) Balancing equations could

Question Video: Describing the Correct Symbol Equation for the Reaction between Zinc Metal and Hydrochloric Acid | Nagwa
![Molecular Zinc Hydride Cations [ZnH]+: Synthesis, Structure, and CO2 Hydrosilylation Catalysis - Ritter - 2020 - Angewandte Chemie International Edition - Wiley Online Library Molecular Zinc Hydride Cations [ZnH]+: Synthesis, Structure, and CO2 Hydrosilylation Catalysis - Ritter - 2020 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/8fb7eef0-0324-42ed-a2b2-135946ab80b4/anie202011480-toc-0001-m.png)
Molecular Zinc Hydride Cations [ZnH]+: Synthesis, Structure, and CO2 Hydrosilylation Catalysis - Ritter - 2020 - Angewandte Chemie International Edition - Wiley Online Library

Hydrosilane σ‐Adduct Intermediates in an Adaptive Zinc‐Catalyzed Cross‐dehydrocoupling of Si−H and O−H Bonds - Patnaik - 2021 - Chemistry – A European Journal - Wiley Online Library